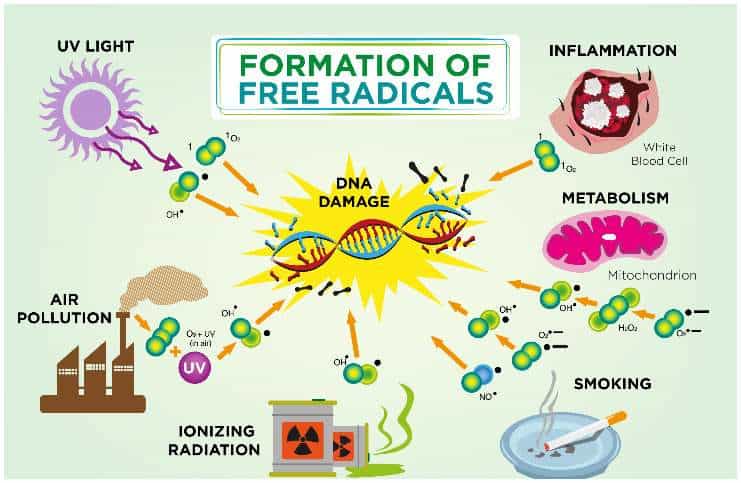
What are free radicals?
They are highly reactive molecules with an odd electron (or “free”) on the external orbital, imbalance condition that turns these fragmented molecules into very unstable and dangerous biochemical agents.
The complex chemical compounds, such as those in the structure of the human being, acquire stability through parity of the molecules’ electrons.
When a molecule loses an electron, it becomes unbalanced in electrochemical terms, biochemically unstable and highly reactive, seeking to regain stability by violently stealing an electron from another molecule.
Whenever a free radical (for example, originating from toxic waste from the environment) attacks a normal molecule to steal its electron, the molecule modifies its initial properties as a result of this process, turning itself into a free radical.
On a molecular level, a mutilated cell loses its basic functions, turning into a source of new free radicals, ready to attack other cells and distorting their functions. Thus, it generates an uncontrolled chain of biochemical reactions that are harmful to the body, with dire repercussions in time.
Oxidation is a chemical reaction during which electrons are transferred by a substance to an oxidizing element. This reaction only occurs in the presence of oxygen. This type of reaction can produce free radicals.
These are chemical agents – oxygen, nitrogen monoxide – which can lead to chain reactions, possibly causing the death of the cells. These elements will attack cells, DNA, certain molecules, proteins, or fatty acids, in order to degrade or destroy them.
Let’s take rust for instance, which is the oxidation of iron, based on oxygen and water. Oxygen is, in conclusion, a highly corrosive element for iron and body as well. They are destructive to the body, although they play an important role in their survival.
Where do they come from?
Internal sources are the processes of metabolic acidosis, triggered in the body due to prolonged stress and accumulation of persistent negative emotions.
External sources include nuclear radiation, X-rays and microwaves, toxic metals such as aluminum and cadmium in drinking water, iron (consumed too much), smog, chemical food additives, cigarette smoke, exhaust gases (especially lead compounds) and, perhaps the most significant, hydrogenated vegetable oils, ever-present in usual products such as margarine.
These artificial fats oxidize the moment they come into contact with air and continue this process inside the body, causing a chain of mutilation reactions on the molecular level, that damage cells and vital functions with a higher speed than that of the body’s defense capacity.
All substances listed above produce free radicals when oxidized (combined with oxygen) and broken down.
Body’s aging process
The role in the cells’ aging process is a hypothesis known as “oxidative stress”, that was defined in 1954 by American professor Denham Harman. The basic principle of this hypothesis is that aging of the cells and body, as well as the associated degenerative diseases, are related to the erosion action of free radical, which attacks body cells.
If we can limit the occurrence of free radicals and counteract the ones present in the body, we could live longer and be healthier.
In addition to the oxidation and as a „bonus” for the destruction of proteins or fatty acids, free radicals attack the cells’ DNA. Fortunately, certain agents produced by the body can repair DNA and counteract free radicals.
But if damages are not fully countered and if DNA’s repair is not complete, cells degrade and change their structure, which happens along with aging. As we age and as the body accumulates more attacks, the number of damaged cells will be greater.
Many diseases are related to cell degeneration
Wrinkles and skin aging: proteins that constitute the skin structure are attacked by free radicals, weakening that structure, thus leading to less resistant skin.
Cataract: as skin proteins, eye lens’ proteins degrade by oxidation and the vision diminishes.
Heart attack: the fatty acids degraded by free radicals are deposited on the walls of blood vessels, leading to poor blood circulation.
Cancer: repeated attacks on the DNA can destroy it and lead to distortions of cells, thus causing cancer.
You should know that the level of free radicals is related to the body’s metabolism. The metabolism defines the activity of the body, the number of calories ingested through eating, the number of calories used by the body, the number of heartbeats or breathing.
The higher the metabolism and more active the body, the greater the production of free radicals that accelerate aging. Under these circumstances, we actually have to find a way to balance. The extremes are always dangerous.
What are the benefits of antioxidants?
Antioxidants – vitamins and minerals – are molecules that prevent oxidation. The oxidation reaction is a chain reaction that antioxidants block, thus preventing free radicals from attacking the body’s cells.
Antioxidants will attach to the free radicals, creating an oxidation reaction with them, which will make them harmless and the radical reaction with proteins or fatty acids impossible.
These antioxidant reactions occur continually in the body and consume a lot of antioxidants. Therefore, we must always ingest them through diet or supplements, in order to allow the destruction of excess free radicals. Thus, there must be a balance between the two elements to stabilize this reaction.
Antioxidants sources:
Allium sulfur – garlic, onion
Anthocyanins – blackcurrants, berries in general, grapefruit, eggplant
Catechins – red wine, green teas, apples, gooseberries, blueberries, grape seeds, strawberries, and kiwis
Beta-carotene – carrots, mangoes, apricots, spinach, parsley
Copper – sunflower seeds, peanuts, lentils, mushrooms, whole grains
Cryptoxanthin – pepper, chili, pumpkin, mango
Flavonoids – green tea, citrus fruits, red wine, onions, apples
Indole – broccoli, cabbage, cauliflower
Isoflavonoids – soy, tofu, lentils
Lignans – sesame seeds, whole grains, flax seeds, vegetables
Lycopene – tomatoes, pink grapefruit, watermelon
Manganese – seafood, whole grains, milk, nuts
Polyphenols – thyme, oregano
Selenium – seafood, whole grains, soy, lentils, beans, garlic, sunflower seeds, cashews, sea buckthorn
Vitamin C – blackcurrants, oranges, lemons, kiwi, mango, spinach, hot pepper, berries
Vitamin E or tocopherol – cold-pressed vegetable oils, avocado, nuts, seeds, whole grains, dark chocolate
Zinc – seafood, pollen, spirulina, nuts.
Take the necessary measures to prevent an excess of free radicals! During the summer protect yourselves from the strong sun, don’t consume excess iron, pay attention to pollution, and embrace a diet based on raw vegetables and fruits.
Thus, you will enjoy a long and healthy life.
References https://www.tandfonline.com/loi/ifra20 http://www.imedpub.com/articles/free-radicals-and-antioxidants-human